Introduction
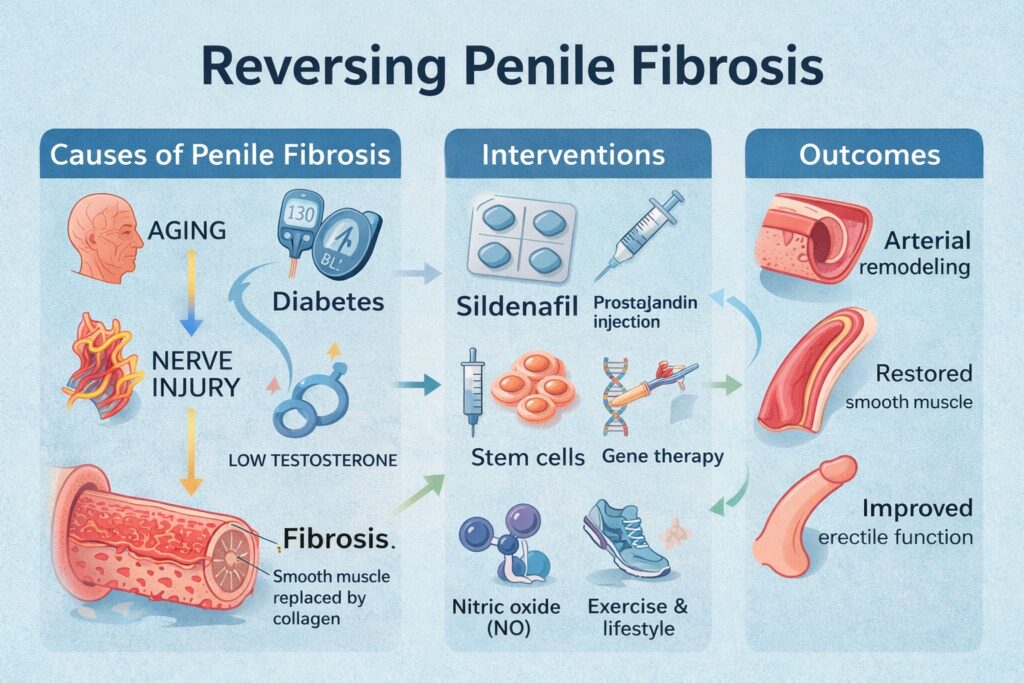
Penile fibrosis represents one of the most complex and underappreciated pathophysiological processes contributing to erectile dysfunction (ED). It lies at the intersection of vascular compromise, smooth muscle apoptosis, hormonal deficiency, oxidative stress, and chronic inflammation. Unlike the transient hemodynamic changes that define common ED, penile fibrosis introduces a permanent structural component—a stiff, inelastic alteration of the corpora cavernosa and tunica albuginea that undermines erectile capacity.
For decades, fibrosis was considered irreversible, a sentence of permanent mechanical dysfunction. However, as our molecular understanding deepens, this dogma is gradually dissolving. Recent research suggests that the process may not only be slowed but in certain conditions partially reversed through targeted pharmacotherapy, regenerative medicine, and lifestyle modulation.
Among these approaches, phosphodiesterase type 5 inhibitors (PDE5Is)—notably sildenafil—and intracavernous prostaglandins such as alprostadil, have emerged as leading contenders. Beyond their well-known erectogenic effects, these agents exert anti-fibrotic, pro-oxygenation, and anti-apoptotic actions at the cellular level. This evolving paradigm reframes erectile pharmacotherapy not merely as symptomatic management but as a potential disease-modifying intervention.
The Pathophysiology of Penile Fibrosis: Where Biology Meets Mechanics
At the microscopic level, penile fibrosis reflects a disruption of the delicate ratio between smooth muscle cells (SMCs) and extracellular matrix (ECM) components, particularly collagen. In the healthy penis, approximately 40–45% of the corpus cavernosum is composed of SMCs, which contract and relax to control penile rigidity. With ageing, diabetes, or nerve injury, SMC apoptosis increases while collagen deposition accelerates, leading to a stiffened, poorly compliant corporal architecture.
The underlying drivers are multifactorial. Chronic hypoxia—whether from atherosclerosis, venous leakage, or post-surgical neuropraxia—stimulates transforming growth factor-β1 (TGF-β1), a key cytokine that promotes collagen synthesis and myofibroblast proliferation. TGF-β1 also suppresses nitric oxide (NO) production, creating a vicious cycle of decreased perfusion and further fibrosis. In parallel, reactive oxygen species (ROS) generated during oxidative stress amplify fibroblast activation and DNA damage.
The tunica albuginea, the fibrous sheath surrounding the corpora, also undergoes degenerative changes. In conditions such as Peyronie’s disease, collagen fibers become abnormally dense and misaligned, losing their elastic interlayer slippage—a structural rigidity that physically distorts the penis during erection. Collectively, these processes culminate in corporal veno-occlusive dysfunction (CVOD), where venous outflow can no longer be effectively trapped, making sustained erection physiologically impossible.
Ageing, Androgen Decline, and the Collagen–Smooth Muscle Imbalance
Ageing remains the single strongest predictor of penile fibrosis. Histological studies reveal that aged corporal tissue exhibits loss of elastic fibers, accumulation of collagen types I and IV, and diminished endothelial nitric oxide synthase (eNOS) expression. The decreased production of NO leads to impaired relaxation of smooth muscle, while chronic hypoxia activates fibroblasts to produce excessive ECM.
The hormonal milieu plays a crucial role. Testosterone is not merely a libido hormone; it is a trophic factor for penile architecture. Androgen deprivation—whether via natural decline or medical castration—leads to atrophy of trabecular smooth muscle, endothelial irregularity, and even adipocyte infiltration in the subtunical region. Animal studies demonstrate that testosterone supplementation reverses these morphological alterations, restoring SMC organization and nitric oxide synthase expression.
Interestingly, the androgen-dependent regulation of vascular progenitor cells may explain why hypogonadism accelerates structural deterioration. Reduced testosterone limits the pool of circulating endothelial progenitors, impairing tissue repair and vascular remodeling. Thus, the ageing penis becomes a biological battlefield of oxidative stress, reduced regenerative potential, and unchecked collagen deposition—a perfect storm for fibrosis.
Diabetes, Nerve Injury, and the Fibrotic Cascade
In diabetes mellitus, fibrosis assumes a more aggressive form. Hyperglycemia triggers advanced glycation end products (AGEs), which in turn stimulate TGF-β1 and connective tissue growth factor (CTGF). The diabetic penis demonstrates generalized fibrosis of the arterial media and loss of SMCs, leading to severe erectile impairment. The Smad signaling pathway, activated downstream of TGF-β1, plays a central role in driving this fibrotic cascade.
Cavernous nerve damage—particularly following radical prostatectomy—acts through a similar mechanism. Denervation deprives corporal tissue of neurogenic nitric oxide release, leading to hypoxia-induced overexpression of hypoxia-inducible factor-1α (HIF-1α) and subsequent fibrosis. Early postoperative loss of nocturnal erections exacerbates this process. Biopsies from post-prostatectomy patients show extensive collagen replacement of corporal SMCs, confirming the structural basis for post-surgical ED.
In both diabetic and neurogenic models, the absence of inducible nitric oxide synthase (iNOS) worsens fibrosis, underscoring the enzyme’s natural anti-fibrotic role. Interestingly, long-term sildenafil administration has been shown to upregulate iNOS and eNOS, countering hypoxia and promoting endothelial repair. Here again, sildenafil’s value extends far beyond symptom relief—it acts as a molecular shield against fibrosis.
The Role of the Nitric Oxide–cGMP Axis: The Foundation of Anti-Fibrotic Therapy
The nitric oxide–cyclic guanosine monophosphate (NO–cGMP) pathway represents the molecular cornerstone of erectile physiology. Nitric oxide, produced by neuronal and endothelial NOS, activates guanylyl cyclase in SMCs, increasing intracellular cGMP and promoting relaxation. Chronic ischemia suppresses this pathway, shifting the balance toward contraction, hypoxia, and ECM accumulation.
Sildenafil, a PDE5 inhibitor, prevents the degradation of cGMP, prolonging its bioactivity. The result is not only improved erectile function but also enhanced oxygen delivery to penile tissue. Studies have shown that long-term administration of sildenafil in aged or diabetic rats reduces collagen content, restores the SMC-to-collagen ratio, and normalizes cavernosal hemodynamics.
Moreover, chronic PDE5 inhibition inhibits the RhoA/Rho-kinase pathway, a signaling cascade implicated in sustained smooth muscle contraction and fibrosis. By blocking Rho kinase activity, sildenafil indirectly maintains corporal compliance and counters the molecular remodeling that drives veno-occlusive dysfunction.
In clinical settings, daily sildenafil or tadalafil following radical prostatectomy has been associated with better preservation of smooth muscle and reduced fibrosis on biopsy, translating into higher rates of spontaneous erectile recovery. Such evidence redefines PDE5 inhibitors as agents of penile preservation, not just performance.
Experimental and Emerging Therapeutic Approaches
The quest to reverse penile fibrosis has spurred innovation across several domains of translational medicine.
1. Gene Therapy:
Gene transfer techniques targeting the NO synthase and Rho-kinase pathways have shown promise. Experimental introduction of iNOS cDNA into fibrotic plaques in Peyronie’s disease models induces collagen regression, while dominant-negative RhoA constructs restore endothelial nitric oxide synthase (eNOS) expression in diabetic rats.
2. Stem Cell Therapy:
Implantation of muscle-derived stem cells into aged rat corpora cavernosa has demonstrated regeneration of smooth muscle tissue and restoration of erectile function. These cells differentiate into both endothelial and SMC lineages, correcting the histological imbalance central to fibrosis. Moreover, pharmacological activation of endogenous stem cells through the NO–cGMP pathway offers a less invasive, future-forward alternative.
3. Antifibrotic Molecules:
Agents that antagonize TGF-β signaling—such as neutralizing antibodies, soluble receptors, or antisense oligonucleotides—show potential to arrest fibrosis progression. Compounds like pentoxifylline, a nonselective phosphodiesterase inhibitor increasing cAMP, demonstrate synergistic effects with sildenafil, reducing collagen expression and improving oxygenation.
Collectively, these innovations suggest that penile fibrosis, once deemed irreversible, might soon be treated through multimodal molecular strategies rather than resigned acceptance.
Peyronie’s Disease and the Tunical Connection
While corporal fibrosis affects erectile function, tunical fibrosis, as seen in Peyronie’s disease (PD), distorts penile anatomy. The tunica albuginea—normally composed of organized, multilayered collagen bundles—loses elasticity and develops noncompliant plaques. The fibrosis in PD is not random; it represents a pathological overactivation of wound healing, driven by TGF-β1, ROS, and inflammatory cytokines.
In PD, inducible nitric oxide synthase (iNOS) acts paradoxically: while inflammation increases its expression, inhibition of iNOS aggravates collagen deposition. Thus, strategies that enhance endogenous nitric oxide signaling, including the use of sildenafil or vardenafil, can mitigate fibrotic plaque progression. Experimental data demonstrate that daily PDE5 inhibitor therapy reduces plaque volume, normalizes collagen subtype ratios, and increases apoptosis of myofibroblasts, all contributing to softening of the tunical lesion.
Furthermore, the interplay between androgen deficiency and PD is increasingly recognized. Low testosterone may predispose to tunical fibrosis by impairing collagen turnover and endothelial resilience. This underscores the value of a hormone-optimized, nitric oxide–enhanced regimen for men at risk.
The Expanding Role of Sildenafil: From Erectile Aid to Anti-Fibrotic Medicine
Sildenafil’s reputation as a lifestyle drug belies its depth as a pharmacological tool. Chronic administration, particularly in low-dose nightly regimens, appears to reverse microstructural changes in penile tissue. Biopsies from men taking sildenafil after prostatectomy revealed significant preservation of smooth muscle and reduction in fibrotic density compared to controls.
Mechanistically, sildenafil interferes with TGF-β1–Smad signaling by preventing nuclear translocation of phosphorylated Smad-2 and Smad-3, key mediators of collagen gene expression. It also maintains endothelial health, reduces oxidative stress, and sustains basal cavernosal perfusion even during non-erect states.
Clinically, early initiation of sildenafil post-surgery shortens recovery time for natural erections. When combined with testosterone replacement in hypogonadal patients or with vacuum erection devices, the outcomes are further enhanced. The future may witness a shift toward chronic low-dose PDE5 inhibition as standard prophylaxis against penile atrophy and fibrosis in men at risk—be it from diabetes, prostate surgery, or vascular disease.
Looking Ahead: Regeneration, Nanomedicine, and Beyond
Beyond pharmacology, the next frontier lies in bioengineering and regenerative delivery systems. Nanoparticle-based carriers for erectogenic agents allow targeted topical delivery, minimizing systemic exposure while maximizing local efficacy. Early animal studies show that nanoparticle formulations of PDE5 inhibitors achieve sustained tissue concentrations and improved erectile response without hepatic metabolism.
Meanwhile, regular aerobic exercise—perhaps the simplest intervention—enhances eNOS and nNOS expression, countering oxidative stress and improving penile perfusion. These lifestyle-driven molecular benefits complement pharmacotherapy and reinforce the principle that rehabilitation is not a single-pill strategy but an integrated regimen.
Gene therapy, stem cell transplantation, and pharmacological modulation of progenitor differentiation collectively point toward a regenerative horizon. If current research continues its trajectory, the distinction between “functional” and “structural” ED may soon blur, replaced by a unified concept of reversible erectile biology.
Conclusion
The reversal of penile fibrosis is no longer a fantasy but a credible scientific goal. By understanding the molecular interplay between hypoxia, TGF-β1, nitric oxide, and smooth muscle dynamics, medicine is gradually transforming from symptomatic treatment toward pathophysiological correction.
Sildenafil, long regarded as a facilitator of erection, now stands recognized as a protector of penile architecture—a pharmacological shield against the molecular erosion of manhood. Its ability to sustain cGMP levels, counteract oxidative stress, and inhibit fibrotic gene expression situates it at the core of modern penile rehabilitation.
Ultimately, the future of sexual medicine will depend on preventive intervention, not crisis response. With a combination of PDE5 inhibition, hormonal optimization, stem cell modulation, and intelligent delivery systems, the aged or injured penis may yet regain its physiological elasticity and vitality. Penile fibrosis, once irreversible, may soon become a manageable—and perhaps reversible—manifestation of systemic vascular health.
FAQ
1. Can penile fibrosis truly be reversed?
Partial reversal is possible. Long-term use of PDE5 inhibitors like sildenafil and vardenafil, combined with good vascular health and hormonal balance, can restore smooth muscle integrity and reduce collagen deposition.
2. Is daily sildenafil safe for chronic use?
Clinical trials and long-term follow-up studies show that daily low-dose sildenafil is generally well tolerated and may improve both erectile and endothelial function when monitored by a physician.
3. Does testosterone therapy help in penile fibrosis?
Yes, particularly in hypogonadal men. Testosterone maintains smooth muscle content, supports nitric oxide synthesis, and prevents adipocyte infiltration of corporal tissue.
4. What lifestyle measures can protect against penile fibrosis?
Regular exercise, smoking cessation, tight glycemic control in diabetics, and avoidance of chronic hypoxia all contribute to maintaining penile elasticity and preventing fibrosis.